General Information
General Information
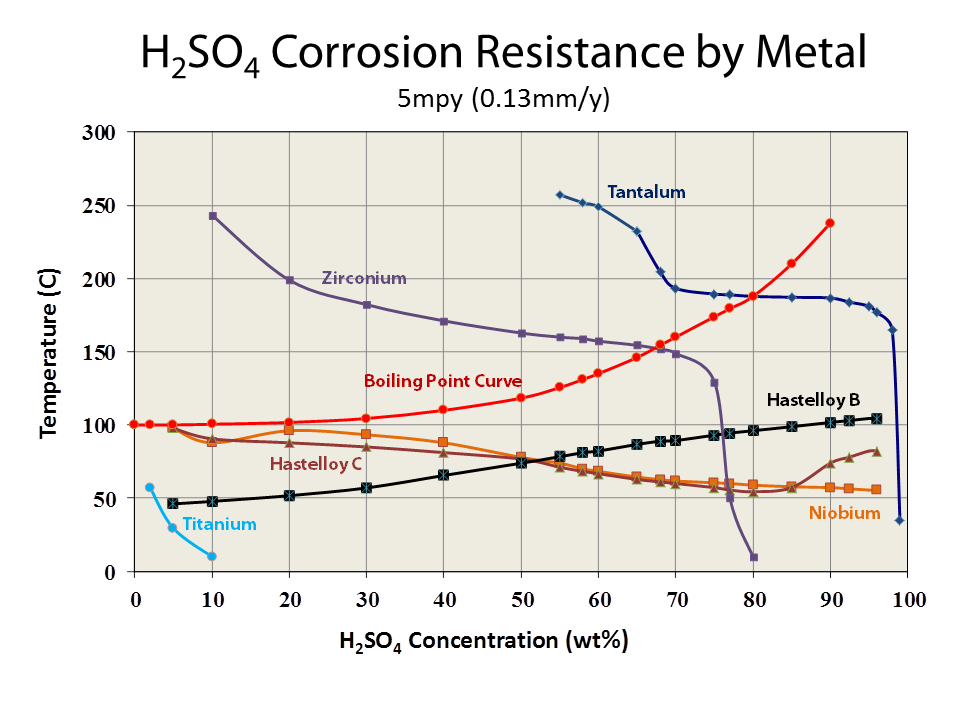
Sulfuric acid (H2SO4) is a very common, strong and yet a strange acid in its behavior. What is unusual about sulfuric acid is that at concentrations over 99% it is not corrosive and could be handled with ordinary materials. However when sulfuric acid is diluted, it becomes very corrosive. This is unusual.
The most common problem with sulfuric acid is the fact that it is extremely exothermic when diluted with water, therefore raising the temperature and the corrosiveness of the solution. Since sulfuric acid is usually shipped and supplied at a 99% concentration this is often the case. As a result, Ultramet CPT treated heat exchangers to chill diluted sulfuric acid is a common application.
Materials
Sulfuric acid with concentrations between 10 and 90% is known to be especially corrosive. Specialty metals like titanium and Hastelloy C-276 will show rapid corrosion in sulfuric acid as low as 120F (50C) for nickel and even lower for titanium. Tantalum metal’s corrosion resistance to sulfuric acid when compared to other materials is 2nd to none.