Basics
Basics
Hydrochloric acid (HCl) is a highly corrosive acid with a variety of industrial uses. HCl is a solution of hydrogen chloride in water and is available in a range of concentrations typically not exceeding reagent grade HCl at 37%. Beyond this concentration HCl fumes and becomes difficult to contain.
Materials Comparison
Polymers: At lower concentration and temperatures, HCl is often contained with fluorinated polymers and polymer linings such as PTFE, PFA and PVDF. These polymers generally perform well at ambient conditions but are susceptible to the HCl vapors that diffuse through the polymer. As the application temperatures increase, the polymers become mechanically soft and the hydrochloric acid becomes exponentially more corrosive. Diffusion through polymers is accelerated, which will lead to polymer swelling and corrosive failures on the backside of the liners. This is a common failure mode for polymers in hydrochloric acid.
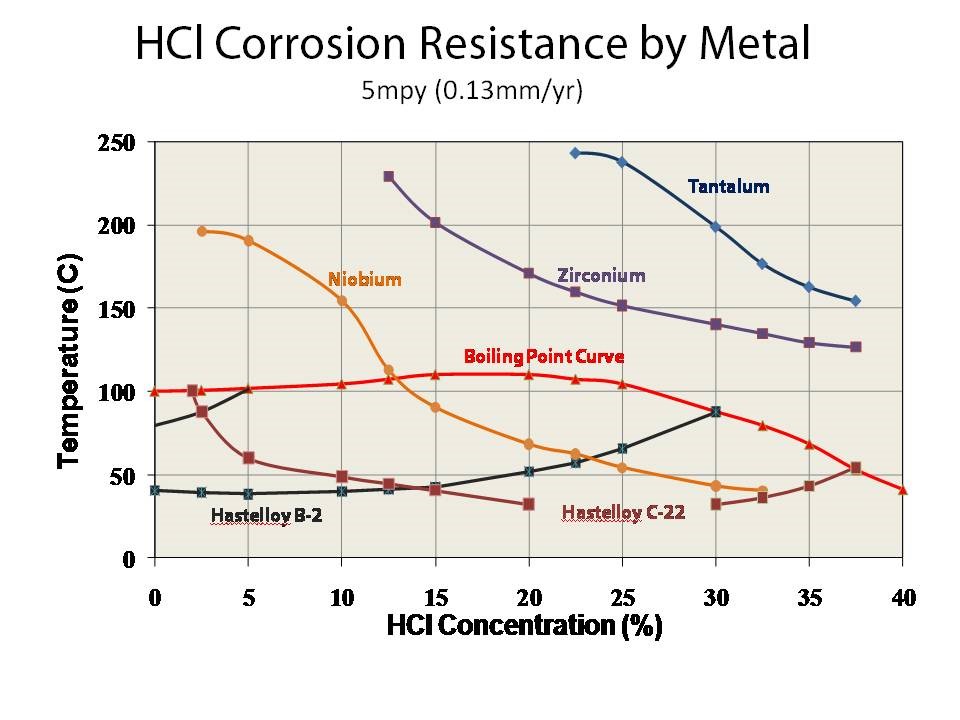
Nickel Alloys: As stainless steel does not provide enough corrosion resistant to hydrochloric acid, typically nickel alloys are deployed in the form of Monel, Inconel or Hastelloy in various grades. Although nickel alloys are far more corrosion resistant than steel to hydrochloric acid it certainly have its limits. The iso-corrosion curve below shows the corrosion resistance of Hastelloy B2 and Hastelloy C22 at various concentration and temperatures. Generally the further below the iso-corrosion lines the better. As we see when looking at the various Hastelloy grades when heatedabove 50C at most concentration of HCl there will be a corrosion rate.
Tantalum Metal: The hydrochloric acid iso-corrosion curves show that tantalum metal has the best corrosion resistance when compared to other specialty alloys at any temperature and concentration including zirconium. For any concentration at temperatures less than 300F or (150C), tantalum has no corrosion rate in hydrochloric acid. This is why tantalum metal is ideal for HCl applications.